Parkinson’s disease has traditionally been primarily associated with a nigrostriatal dopamine deficit resulting in the characteristic motor symptoms of tremor, rigidity, and bradykinesia. Nowadays, the involvement of other brain circuits is widely recognized, as the majority of patients also present numerous non-motor symptoms such as dementia, depression, sleep disorders, or apathy.
 Nevertheless, the mechanistic basis for neuropathological and symptomatic heterogeneity remains unclear. Indeed differential neurotransmitter and receptor expression may underpin the selective vulnerability of several neuronal populations. So the lack of whole-brain spatial distribution maps of neurotransmitter receptors in patients with PD hampers research progress.
Nevertheless, the mechanistic basis for neuropathological and symptomatic heterogeneity remains unclear. Indeed differential neurotransmitter and receptor expression may underpin the selective vulnerability of several neuronal populations. So the lack of whole-brain spatial distribution maps of neurotransmitter receptors in patients with PD hampers research progress.
In a new medRXiv publication, scientists from Canada use patient-specific generative brain modeling to identify neurotransmitter receptor-mediated mechanisms involved in PD progression.
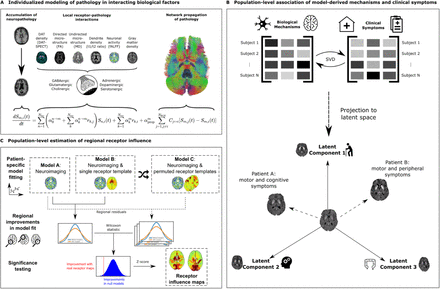
Multifactorial causal modeling (MCM) is a mechanistic modeling approach that is able to identify contributions of interacting factors to longitudinal changes. Combining multi-modal neuroimaging with spatial distribution templates of 15 neurotransmitter receptors from post-mortem autoradiography in an MCM-based approach may significantly improve the explanation of degenerative changes in individual patients’ neuroimaging data, and linked specific receptor-pathology interactions to clinical symptoms.
Thus the authors combined receptor maps with longitudinal neuroimaging (PPMI data), to detect a diverse set of receptors influencing gray matter atrophy, microstructural degeneration, and dendrite loss in PD. Importantly, identified receptor mechanisms correlate with symptomatic variability along two distinct axes, representing motor/psychomotor symptoms with large GABAergic contributions, and cholinergically-driven visuospatial dysfunction.
Furthermore, the authors map cortical and subcortical regions where receptors exert significant influence on neurodegeneration.
Their work constitutes the first personalized causal model linking the progression of multi-factorial brain reorganization in PD across spatial scales, including molecular systems, accumulation of neuropathology in macroscopic brain regions, and clinical phenotypes.
